Researchers at the Indian Institute of Science (IISc) have developed a microparticle formulation that allows slow release of drugs to treat osteoarthritis, a chronic joint condition.
Doctoral student Kaamini Dhanabalan of the Centre for BioSystems Science and Engineering (BSSE) at IISC, guided by Assistant Professor Rachit Agarwal, has designed a polymer matrix made of poly (lactic-co-glycolic acid) or PLGA, an FDA-approved biomaterial, to encapsulate rapamycin, an immunosuppressant drug. Preliminary studies on cells cultured in the laboratory as well as in mice models have shown promising results indicating reduced inflammation and cartilage repair due to sustained drug release.
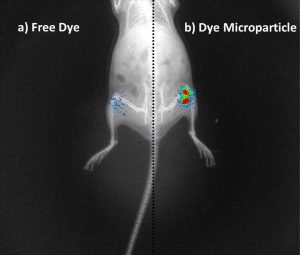
“In cell studies, rapamycin-loaded PLGA microparticles could release the drug for up to 21 days, and in animal studies, PLGA microparticles showed residence time up to 30 days after injecting the microparticles in the mice knee joint,” says Kaamini Dhanabalan who is first author of the study published in the journal Biomaterials Science.
Osteoarthritis (OA) is a joint disease characterized by progressive damage of articular cartilage and the adjoining subchondral bone. This disease happens because of wear and tear of the cartilage ‒ the smooth tissue that protects bone joints ‒ caused due to stress or aging. Chondrocytes, the primary cells of the cartilage, have limited regenerative capacity and when they undergo stress due to trauma or with aging, they senesce or become apoptotic.
The current treatments in vogue revolve more around managing pain and inflammation than treating the disease. Although several classes of drugs seemed promising in preclinical trials, low drug retention and rapid clearance from the target site have made clinical translation difficult.
Rapamycin, a potent immunomodulator, has shown promise in OA treatment. It activates autophagy and is known to prevent senescence. However, its clinical translation for OA is hampered due to systemic toxicity as high and frequent doses are required. Here, we have fabricated rapamycin encapsulated poly(lactic-co-glycolic acid) (PLGA) based carriers that induced autophagy and prevented cellular senescence in human chondrocytes. The microparticle (MP) delivery system showed sustained release of the drug for several weeks.
The formulation developed by the IISc team, however, has a residence time of up to 30 days at the target site, with no evident signs that it may cause discomfort to patients. Such a sustained release system can improve patient compliance and reduce hospital visits.
PLGA is widely used for drug delivery applications and several drug formulations are currently used in clinics. Rapamycin is commonly used to suppress immune response in patients undergoing surgery for organ transplant to prevent organ rejection. Preclinical studies have shown its potential for treating osteoarthritis by preventing cell loss and cartilage damage, thereby reducing inflammation. However, the short drug retention time of ~1-4 hours demands frequent injections to maintain the therapeutic window in the joints. Therefore, Dhanabalan and her colleagues combined the advantages of PLGA and rapamycin to create a system that would allow sustained release of the drug. This was achieved by encapsulating rapamycin in PLGA microparticles.
To evaluate the effectiveness of this formulation, chondrocytes or cartilage cells were cultured and subjected to various stresses to recreate osteoarthritis-like conditions under laboratory settings. This resulted in ailing chondrocytes with the hallmarks of osteoarthritis. These damaged chondrocytes recovered from osteoarthritis when treated with rapamycin-loaded PLGA microparticles.
“Preliminary studies using this newly-designed formulation can potentially reduce the frequent medical intervention to once a month. Detailed studies are in progress to explore the functional potential in mouse models of osteoarthritis,” says Rachit Agarwal, Assistant Professor at BSSE, IISc, and senior author of the study.
Also read: Anti-ageing sunscreens from prawn shells